Ultraviolet light ("black light") - broad
spectrum (may be available from a
high school science laboratory or a
rock and mineral collector)
Fluorescent mineral (See note in the
discussion section.)
Window glass
Dark room
Procedure:
1. Darken the room and turn on the ultraviolet (UV) light. (See Caution at the end of the discussion section.) Direct the UV light's beam onto the fluorescent minerals. Observe the color of the emitted light.
2. Place the glass between the light and the mineral and again observe the emitted light.
Discussion:
Certain minerals and a variety of other substances fluoresce or emit visible light when illuminated by ultraviolet light. Fluorescence is a process that exchanges ultraviolet-light energy for visible-light energy. Photons of ultraviolet light are captured by electrons orbiting the nuclei of atoms within those materials. The electrons, gaining energy, are boosted to excited energy states. The electrons eventually release this captured energy as visible light as they return to lower energy states.
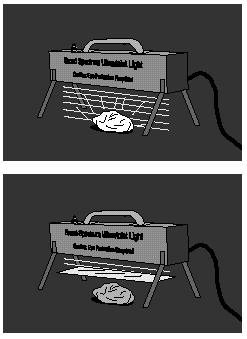
Low-energy ultraviolet light--sometimes called long-wave UV--penetrates to Earth's surface. This low-energy ultraviolet light causes Day-Glo paints to give off spectacular colors and white clothing to glow brightly when washed in detergents that contain fluorescent dyes (advertised as making clothes "whiter than white"). Most of the high-energy ultraviolet light--sometimes called short-wave UV--is blocked by the ozone layer in Earth's upper atmosphere. Higher-energy ultraviolet light causes skin
